Testosterone Optimization in Males for Healthspan Extension
Low testosterone is not exclusively tied to age-related declines. Secondary and functional hypogonadism are frequently driven by obesity, insulin resistance, sleep disruption, and chronic stress. Addressing these upstream factors is foundational to sustainable hormone optimization.
Therapy selection should match physiology and goals. Exogenous TRT reliably raises testosterone but suppresses endogenous production and fertility. Endogenous stimulation strategies preserve axis function and may better align with long-term healthspan goals in appropriately selected men.
Enclomiphene restores rather than replaces testosterone. By selectively stimulating the hypothalamic–pituitary–gonadal axis, enclomiphene increases LH and endogenous testosterone often by 200–300 ng/dL while maintaining spermatogenesis and avoiding undesirable side effects.
Monitoring matters as much as dosing. Responsible optimization includes tracking total and free testosterone, LH, estradiol, hematocrit, SHBG, and metabolic markers to ensure physiologic restoration rather than unchecked hormone escalation.
Testosterone optimization is not about supraphysiologic enhancement; it is about restoring mid-range physiologic levels that support muscle mass, metabolic flexibility, bone density, cognitive resilience, and long-term vitality.
Introduction
Over the past several decades, average testosterone levels in men have declined at a population level, independent of normal aging. Contemporary analyses show that men today have significantly lower serum testosterone concentrations compared to age-matched men from previous generations (Figure 1), even after adjusting for body mass index and comorbid conditions [1,2]. Since the 1970s, for example, average testosterone levels have declined by nearly 200 ng/dl from 22 nmol/l (635 ng/dl) to under 15 nmol/l (433 ng/dl) [1]. This downward trend has been documented across multiple cohorts and is increasingly attributed to a convergence of metabolic, environmental, and lifestyle factors.
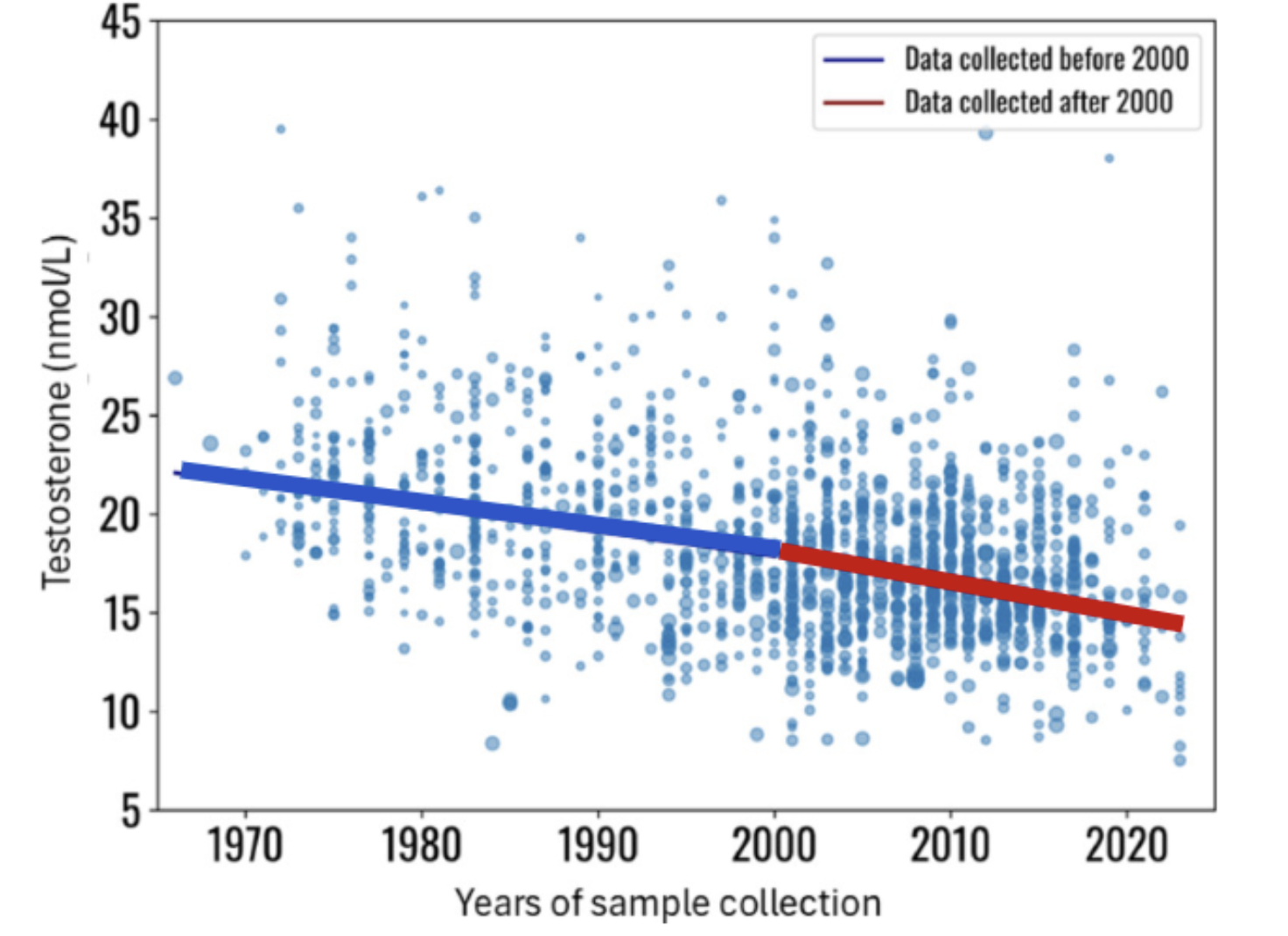
Figure 1. Temporal Testosterone concentration by decade.
As baseline testosterone levels have shifted downward, clinical recognition of hypogonadism, particularly secondary and functional forms, has expanded. In parallel, the use of testosterone optimization therapies has risen dramatically, increasing nearly fourfold in recent years [3]. This growth reflects both heightened awareness of testosterone’s role in metabolic, musculoskeletal, and cognitive health and a promising cultural shift toward proactive healthspan management.
The convergence of declining endogenous testosterone and rising therapeutic use presents a critical inflection point. The modern question is no longer whether testosterone matters for long-term health; it clearly does, but how best to restore and optimize levels safely, physiologically, and in alignment with long-term endocrine integrity.
Why Hormone Optimization Matters for Male Longevity
Testosterone is often framed narrowly as a reproductive hormone. In reality, it functions as a systems-level regulator of metabolism, tissue maintenance, and physiological resilience. Beyond its role in sexual development, testosterone influences lean mass preservation, fat distribution, insulin sensitivity, erythropoiesis, bone remodeling, vascular tone, mood, and aspects of cognitive function [4–8]. As such, age-related declines in testosterone are not merely cosmetic or sexual; they may reflect broader shifts in anabolic balance that influence healthspan.
Multiple longitudinal cohort studies demonstrate that men in the lowest endogenous testosterone quartiles exhibit significantly higher rates of all-cause mortality, cardiovascular events, metabolic syndrome, frailty, and depression compared with men in mid-physiologic ranges even after adjustment for age and comorbidities [4–7]. While observational data cannot establish causality, the consistency of these associations across diverse populations suggests that testosterone status functions as a meaningful biomarker of systemic health.
Interventional evidence has further shaped the field. In hypogonadal men, normalization of testosterone levels is associated with reductions in fat mass, improvements in glycemic control, increased lean mass and strength, and favorable shifts in cardiometabolic markers [8]. Importantly, the large TRAVERSE trial, designed specifically to evaluate cardiovascular safety, demonstrated that appropriately prescribed testosterone therapy did not increase major adverse cardiovascular events in men with hypogonadism [9]. This finding has significantly reframed prior safety concerns and contributed to the current inflection point in clinical thinking.
Restoring deficient levels to a stable, optimized, physiologic range can support metabolic and musculoskeletal resilience as men age. In the sections that follow, we examine the biology of testosterone decline, the clinical evidence surrounding replacement strategies, and the emerging role of selective estrogen receptor modulation (SERM), particularly enclomiphene, which provides a physiologically aligned approach to long-term testosterone optimization with minimal side effects.
Hypogonadism Types and Healthspan Impacts
Male hypogonadism is often described simply as “low testosterone.” But biologically, it is not a single disease. It is a clinical syndrome that can arise from disruptions at different points along what is known as the hypothalamic–pituitary–gonadal (HPG) axis—the hormonal network that regulates testosterone production [7,8].
Understanding where the disruption occurs is critical. Each location along this axis represents a different underlying biological problem—and therefore may require a different therapeutic approach.
The HPG axis functions as a relay system. The hypothalamus, a small region deep within the brain, initiates the process by releasing gonadotropin-releasing hormone (GnRH). This signal travels to the pituitary gland, which responds by secreting luteinizing hormone (LH) and follicle-stimulating hormone (FSH). These hormones then travel through the bloodstream to the testes, where they stimulate testosterone production and sperm development.
When this system functions properly, it operates through tight feedback loops. Testosterone levels signal back to the brain, helping regulate how much GnRH and LH are released. It is a dynamic, constantly adjusting circuit.
Hypogonadism can develop when this system breaks down at different levels. (Figure 2).
Primary hypogonadism originates within the testes themselves. In this case, the testes are unable to produce adequate testosterone even though the brain and pituitary are sending appropriate signals. Causes may include genetic conditions, prior chemotherapy, infection, or direct testicular injury. Because the signaling from the brain remains intact, LH and FSH levels are often elevated as the body attempts to compensate.
Secondary hypogonadism, by contrast, begins upstream. Here, the hypothalamus or pituitary fails to produce sufficient GnRH, LH, or FSH. As a result, the testes receive inadequate stimulation despite being structurally capable of producing testosterone. In this scenario, hormone levels from the brain are low or inappropriately normal, and testosterone production falls as a consequence.
A third and increasingly common category, functional hypogonadism, reflects disruption of hormonal regulation in the absence of structural damage. Here, age-associated conditions such as obesity, insulin resistance, type 2 diabetes, chronic inflammation, and sleep disturbance alter HPG axis signaling and suppress endogenous testosterone production.
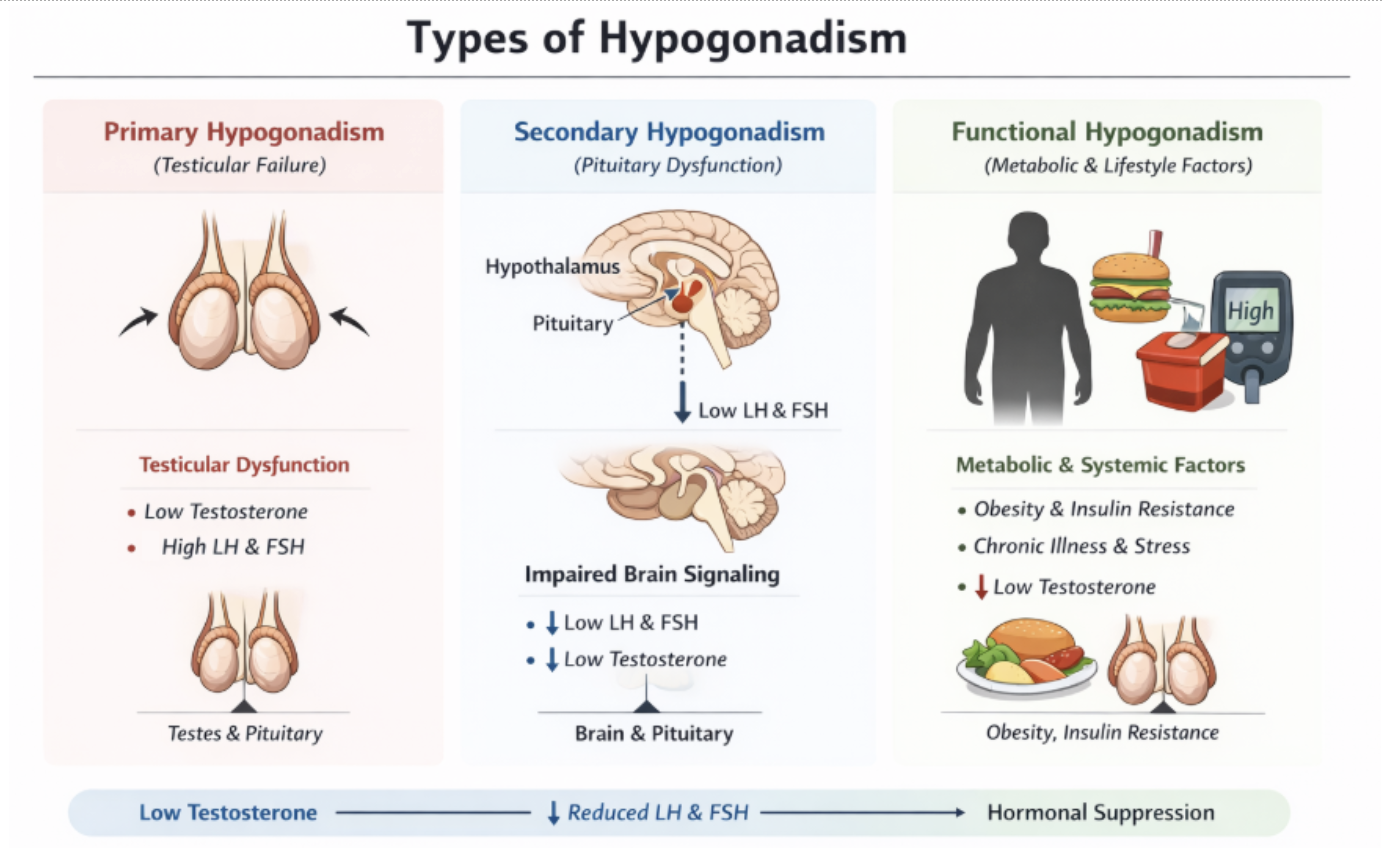
Figure 2. Mechanistic origins of primary, secondary, and functional hypogonadism.
Regardless of subtype, sustained testosterone deficiency produces a constellation of symptoms that extend far beyond reproductive function. Clinically, men often present with fatigue, depressed mood, reduced cognitive clarity, diminished libido, and erectile dysfunction. Physiologically, testosterone deficiency contributes to increased visceral adiposity, loss of lean mass, reduced bone density, impaired erythropoiesis, and metabolic dysregulation; all factors that accelerate frailty and diminish long-term resilience [7].
Importantly, aging alone is rarely the sole driver of hypogonadism. More often, systemic metabolic stress acts as the catalyst. Excess adiposity increases aromatization of testosterone to estradiol; insulin resistance and inflammatory cytokines suppress GnRH signaling; sleep fragmentation impairs nocturnal testosterone pulses. The result can become self-reinforcing: declining testosterone worsens body composition and metabolic health, which in turn further suppresses endogenous production.
These distinctions carry direct relevance for healthspan-oriented therapy. Men with primary hypogonadism typically require direct androgen replacement to restore physiologic levels. In contrast, men with functional or secondary forms may benefit from strategies that restore endogenous production either through metabolic correction, targeted pharmacologic stimulation of the HPG axis, or both. Recognizing which mechanism predominates allows clinicians to move beyond treating a laboratory value and toward addressing the biological drivers of hormonal decline.
A Brief History: From Testosterone Replacement to Restoration
For decades, testosterone replacement therapy, TRT, has been the standard treatment for classical hypogonadism. Injectable and transdermal formulations reliably elevate serum testosterone and improve symptoms, particularly in men with primary testicular failure [7]. Mechanistically, TRT is more simple; exogenous testosterone replaces circulating levels directly.
However, this approach carries physiologic trade-offs. Through negative feedback, exogenous testosterone suppresses luteinizing hormone (LH) and shuts down endogenous testosterone production. Over time, this can reduce intratesticular testosterone, impair spermatogenesis, and compromise fertility [7,8]. Increased aromatization may also elevate estradiol in susceptible individuals [10].
For men with irreversible primary hypogonadism, these effects may be acceptable. But for younger men, men who maintain the ability to produce testosterone naturally (secondary or functional hypogonadism), or those prioritizing fertility, the endogenous suppression of testosterone production with TRT may be undesirable.
This reality has reshaped how clinicians and patients think about testosterone optimization. The goal is no longer limited to replacing hormone levels when they fall, but to restore physiologic production when possible [7,8,10]. Modern testosterone optimization now spans a spectrum of strategies, ranging from exogenous replacement for men with structural testicular failure to endogenous restoration approaches that stimulate the HPG axis in men who retain functional capacity. Rather than a one-size-fits-all model, contemporary care recognizes that testosterone optimization should align with an individual’s underlying biology, fertility goals, and metabolic profile in addition to providing consistent monitoring and clinical support.
Testosterone Optimization Strategies
The growing recognition that testosterone influences metabolic, musculoskeletal, and cognitive resilience has led to multiple therapeutic strategies aimed at restoring physiologic androgen levels. These approaches differ not only in mechanism but in how they interact with the HPG axis, fertility, long-term monitoring requirements, and suitability for functional versus structural hypogonadism. Rather than viewing these options as interchangeable, it is more beneficial to understand each strategy through the lens of mechanism, magnitude, and healthspan alignment. In this section, we will outline the historical development, physiologic rationale, and practical trade-offs of the major testosterone optimization strategies.
Testosterone Replacement Therapy (TRT)
Testosterone replacement therapy, TRT, emerged in the early 20th century following the isolation and synthesis of testosterone in the 1930s [11]. It was initially reserved for men with clear primary testicular failure, such as Klinefelter syndrome, traumatic injury, infection, or chemotherapy-induced damage, where endogenous production was irreversibly impaired despite intact hypothalamic and pituitary signaling [6].
With the development of injectable esters and transdermal formulations, TRT became the standard treatment for symptomatic androgen deficiency [11,12]. Mechanistically, it bypasses the testes and directly restores circulating testosterone. For men with severe hypogonadism, particularly those with total testosterone levels below 200 ng/dL, TRT remains the most reliable and potent method of normalization [7,8]. It can consistently raise serum levels into the mid- to upper-physiologic range, approximately 600 to 800 ng/dL, and, if dosed more aggressively, can exceed physiologic thresholds altogether.
This potency is both its advantage and its limitation. Unlike endogenous stimulation strategies, TRT does not depend on pituitary reserve or testicular responsiveness; its effect is dose dependent. In clinical practice, the aim is physiologic restoration. Outside supervision, however, supraphysiologic levels significantly above 1,000 ng/dL (35 nmol/L) are achievable, as seen in performance or aesthetic-oriented settings, such as sports or physique competition [7]. From a healthspan perspective, the objective is firmly focused on restoration, not excess, which harms longevity.
The trade-off is HPG axis suppression. Exogenous testosterone reduces luteinizing hormone (LH) through negative feedback, lowering endogenous production and intratesticular testosterone. Over time, this can impair spermatogenesis and fertility. TRT may also increase estradiol through aromatization and elevate hematocrit, necessitating structured monitoring.
For men with primary, structural, hypogonadism, TRT to raise testosterone to physiologic levels provides a wide range of benefits, including better vascular function, mood, muscle strength, bone density, and sexual health in healthy men [7,10]. However, in men with sufficient testicular function, particularly those with secondary or functional hypogonadism, the question becomes whether such potency is necessary, or whether endogenous restoration offers a more physiologically aligned long-term strategy that maintains fertility.
Testosterone Restoration Strategies
For men with intact testicular function, preserving endogenous signaling often becomes the preferred objective. This distinction has given rise to endogenous hormone restoration strategies, which stimulate the body’s own testosterone production rather than replacing it. SERMs, including clomiphene, enclomiphene, and tamoxifen, increase luteinizing hormone (LH) through central feedback modulation, while human chorionic gonadotropin, hCG, mimics LH directly at the testicular level.
Although these approaches typically produce more moderate increases than TRT, the effect is clinically meaningful. In appropriately selected men, testosterone often rises by 150 to 300 ng/dL, restoring levels from the low 300s into the mid-physiologic range [13]. Importantly, these strategies preserve endogenous production and spermatogenesis, offering a physiologically aligned pathway for men with secondary or functional hypogonadism who seek optimization without lifelong axis suppression.
Human Chorionic Gonadotropin (hCG)
Human chorionic gonadotropin (hCG) occupies a distinct place among testosterone restoration strategies. Biologically, it closely resembles luteinizing hormone (LH)—the signal normally released by the pituitary gland to stimulate testosterone production. Because of this structural similarity, hCG can bind directly to LH receptors in the testes and activate Leydig cells, which are responsible for producing testosterone.
In practical terms, hCG acts as a direct stimulant of the testes. Unlike selective estrogen receptor modulators (SERMs), which work upstream by encouraging the brain to release more LH, hCG bypasses the hypothalamus and pituitary entirely. It delivers the signal directly at the testicular level.
Clinically, this makes hCG particularly useful in fertility-preserving strategies. When men receive exogenous testosterone replacement therapy (TRT), natural LH production declines through negative feedback, which can reduce intratesticular testosterone and lead to testicular atrophy. Adding hCG can maintain intratesticular testosterone levels and help preserve spermatogenesis.
As a standalone therapy, hCG can meaningfully raise serum testosterone. In one study, testosterone levels increased by nearly 50%—from 362 ng/dL to 520 ng/dL—over approximately eight months. This confirms that directly stimulating the testes can restore physiologic production in appropriately selected patients [14].
Despite its effectiveness, hCG is not typically a first-line strategy for routine optimization. It requires injections, may elevate estradiol through aromatization, and is generally less convenient than oral SERMs such as enclomiphene. As such, it is best viewed as a specialized tool rather than a foundational approach. This distinction naturally shifts attention toward strategies that recalibrate central feedback mechanisms, bringing the focus to selective estrogen receptor modulation and, ultimately, to enclomiphene.
Tamoxifen
Tamoxifen is one of the earliest SERMs, originally developed in the 1960s and widely used in estrogen receptor positive breast cancer [15]. By antagonizing estrogen receptors in a tissue-selective manner, it reduces estrogen-mediated feedback at the hypothalamus and pituitary, increasing luteinizing hormone and stimulating endogenous testosterone production [16]. This mechanism parallels that of clomiphene and enclomiphene and led to its off-label use in men with gynecomastia, infertility, and secondary hypogonadism.
However, tamoxifen is not commonly used as a primary strategy for long-term testosterone optimization. Its broader tissue effects and oncology-focused pharmacology make it less targeted than newer agents. Clinically, it is most useful for treating gynecomastia or mitigating estrogen-related side effects, and in select fertility protocols [16]. For sustained testosterone restoration in men with secondary or functional hypogonadism, more refined hypothalamic modulators such as enclomiphene are generally preferred. Tamoxifen, therefore, occupies a specialized, situational role rather than serving as a foundational healthspan strategy.
Clomiphene Citrate (Clomid)
Clomiphene citrate was originally developed in the 1950s as a treatment for female infertility. It belongs to a class of medications known as selective estrogen receptor modulators (SERMs). Chemically, clomiphene is composed of two isomers—enclomiphene and zuclomiphene—each with distinct biological effects [13].
Its relevance in men lies in how it influences the hypothalamic–pituitary–gonadal (HPG) axis. Under normal conditions, circulating estrogen provides negative feedback to the hypothalamus and pituitary, signaling that sufficient sex hormone levels are present. Clomiphene partially blocks this estrogen-mediated feedback. As a result, the brain interprets hormone levels as lower than they actually are and increases production of gonadotropin-releasing hormone (GnRH), luteinizing hormone (LH), and follicle-stimulating hormone (FSH).
The rise in LH then stimulates the testes to produce more testosterone. In this way, clomiphene does not replace testosterone—it prompts the body to produce more of its own [13,17].
This distinction is important. Unlike testosterone replacement therapy, which can suppress natural LH production and impair sperm production through feedback inhibition, clomiphene amplifies existing HPG signaling while preserving spermatogenesis.
For men seeking to increase testosterone without compromising fertility, this mechanism makes SERMs an attractive therapeutic category.
Clinically, clomiphene is most effective in men with secondary or functional hypogonadism, where central signaling remains intact but suppressed, such as in obesity, insulin resistance, or age-related metabolic dysfunction. It is not effective in primary hypogonadism, where testicular responsiveness is impaired.
While clomiphene reliably increases testosterone and maintains fertility, its dual-isomer composition introduces pharmacologic complexity [13,17]. The zuclomiphene component has partial estrogenic activity and a longer half-life, which may contribute to mood changes, visual disturbances, and other side effects in some men. This limitation prompted refinement of the SERM strategy, narrowing focus to the purified trans-isomer, enclomiphene, as a more targeted and potentially better-tolerated approach to long-term testosterone restoration.
Enclomiphene
Enclomiphene citrate is the purified trans-isomer of clomiphene and represents a more targeted evolution of selective estrogen receptor modulation for men with hypogonadism.
As discussed earlier, clomiphene citrate contains two stereoisomers: enclomiphene and zuclomiphene. Although they are chemically similar, their biological effects differ. Enclomiphene acts primarily as an anti-estrogen at the level of the hypothalamus and pituitary, blocking estrogen’s negative feedback and stimulating the release of luteinizing hormone (LH). Zuclomiphene, by contrast, has partial estrogen-like activity and a longer half-life, meaning it can remain in the body longer and exert more prolonged hormonal effects [13].
Over time, clinical experience showed that while clomiphene reliably increases testosterone in men, some adverse effects—such as mood changes or visual disturbances—were thought to be related, at least in part, to the estrogenic activity and persistence of the zuclomiphene isomer.
Enclomiphene was therefore isolated as a more refined therapeutic strategy. By removing the isomer with partial estrogen agonist activity, enclomiphene aims to preserve the testosterone-stimulating effects—through increased LH and FSH—while minimizing prolonged estrogen receptor activation.
In essence, enclomiphene seeks to maintain the central mechanism of clomiphene—restoring endogenous testosterone production through HPG axis stimulation—but in a more selective and potentially better-tolerated form [13,17].
Mechanistically, enclomiphene selectively antagonizes estrogen receptors at the hypothalamus, reducing negative feedback and increasing gonadotropin-releasing hormone (GnRH), luteinizing hormone (LH), and follicle-stimulating hormone (FSH) [13]. This upstream stimulation enhances endogenous testosterone production without suppressing the HPG axis. Importantly, because it preserves intratesticular testosterone and spermatogenesis, enclomiphene maintains fertility, an important distinction from exogenous testosterone therapy.
In clinical studies, enclomiphene has been shown to increase total testosterone by approximately 200–300 ng/dL over 4–12 weeks in appropriately selected men [13,18,19]. Practically, a baseline level in the low 300s may rise into the mid-physiologic range (often 600–650 ng/dL), assuming intact pituitary signaling and testicular responsiveness. Because its efficacy depends on functional Leydig cells, enclomiphene is best suited for men with secondary or functional hypogonadism rather than primary testicular failure.
Taken together, enclomiphene occupies a distinct therapeutic niche: it supports physiologic restoration rather than pharmacologic replacement, offering a strategy for testosterone optimization that preserves endogenous endocrine architecture while avoiding the axis suppression associated with traditional TRT.
Among the available testosterone restoration options available to date, enclomiphene occupies a particularly compelling position for healthspan-focused care: it restores physiologic testosterone while preserving endogenous architecture, fertility, and limits so-called estrogenic side effects.
Enclomiphene: Magnitude and Clinical Relevance of Effect
One of the most clinically compelling aspects of enclomiphene is not simply that it raises testosterone but that it does so meaningfully and predictably within physiologic bounds. In controlled trials, enclomiphene has been shown to increase total testosterone by approximately 200–300 ng/dL over several weeks in appropriately selected men [13,18,19]. For individuals beginning in the low hypogonadal range (e.g., 300–350 ng/dL), this often translates into restoration to the mid-normal physiologic range (approximately 550–700 ng/dL), assuming intact pituitary signaling and adequate testicular responsiveness.
This magnitude of increase is clinically significant. A shift from the low 300s into the 600s is not merely cosmetic on a lab report; it represents a transition from symptomatic deficiency to biochemical normalization in many men. Improvements in energy, libido, resistance training response, body composition, and overall vitality frequently parallel this biochemical restoration when lifestyle factors are also aligned. In the appropriate clinical context, enclomiphene’s effect size approaches that of moderate-dose TRT but without HPG axis suppression.
Timeline of Expected Enclomiphene Effects
The tempo of response follows a predictable biological sequence:
Weeks 2–4
LH rises first, reflecting hypothalamic–pituitary engagement. Total testosterone begins increasing shortly thereafter. Many men report early improvements in morning energy, libido, and mental clarity during this window.
Weeks 6–8
As testosterone stabilizes in the mid-physiologic range, improvements in mood, motivation, and exercise recovery often become more noticeable. Gym performance may improve, particularly in resistance-trained individuals.
8-12 Weeks and Beyond
By three months, hormonal stabilization is typically achieved. When paired with consistent training, sleep optimization, and improved nutrition, measurable shifts in body composition, including increased lean mass and reductions in visceral adiposity, may begin to emerge. Some individuals also demonstrate improvements in fasting glucose or insulin sensitivity, particularly when baseline metabolic dysfunction was present.
After 3 months, hormone levels often stabilize, at which point follow-up lab work enables dose adjustments based on symptom trajectory and biomarker trends.
Does Enclomiphene Suppress Natural Testosterone Production?
A common concern is whether enclomiphene suppresses endogenous testosterone production in the way exogenous testosterone replacement therapy does. Mechanistically, it does not. During active treatment, enclomiphene reduces estrogen-mediated negative feedback at the hypothalamus, increasing gonadotropin-releasing hormone and luteinizing hormone [13]. The rise in luteinizing hormone stimulates the testes to produce more endogenous testosterone. Rather than bypassing the HPG axis, enclomiphene amplifies its signaling while preserving intratesticular testosterone and spermatogenesis.
After discontinuation, the pharmacologic stimulus fades. Estrogen feedback resumes its normal regulatory role, luteinizing hormone declines toward baseline, and testosterone levels typically return toward pre-treatment values over several weeks to months. Current evidence does not suggest that enclomiphene permanently resets or reprograms the HPG axis [13,17]. However, context matters. If therapy coincides with meaningful improvements in body composition, sleep, stress, and metabolic health, testosterone may stabilize at a higher level than baseline [20]. In that case, sustained benefit reflects improved physiology rather than a lasting drug effect. Ultimately, enclomiphene leverages natural production, but long-term durability depends on the health of the underlying endocrine terrain.
Comparison and Clinical Decision Making
Choosing a testosterone optimization strategy is less about identifying a universally “best” therapy and more about aligning treatment with physiology, goals, and long-term health trajectory. Exogenous testosterone replacement therapy, TRT, provides predictable and often robust increases in serum testosterone and is well suited for men with primary hypogonadism or structural testicular failure [7]. However, by suppressing luteinizing hormone, it reduces endogenous production and intratesticular testosterone, which may impair fertility and requires ongoing monitoring of hematocrit and estradiol.
Endogenous restoration strategies operate differently. SERMs increase luteinizing hormone by reducing estrogen-mediated feedback, stimulating the body’s own testosterone production [13]. Enclomiphene, as a purified isomer, offers more targeted HPG axis stimulation, while hCG acts directly at the testicular level. Testosterone restoration typically produces moderate but clinically meaningful increases while preserving spermatogenesis, making them particularly attractive for men with secondary and/or functional hypogonadism.
Baseline biomarkers help guide selection. Low testosterone with elevated luteinizing hormone suggests primary hypogonadism and often favors replacement. Low testosterone with low or normal luteinizing hormone suggests secondary or functional suppression, where restoration strategies may be more appropriate. Additional markers, including SHBG, estradiol, hematocrit, metabolic profile, and fertility goals, further refine decision-making for a personalized care plan.
Most importantly, testosterone optimization should occur within a structured clinical partnership. Laboratory interpretation, dose titration, and monitoring of hematologic and metabolic markers are essential for safety and durability. Lifestyle interventions, including resistance training, sleep optimization, and visceral fat reduction, enhance responsiveness and may allow for lower doses over time, in addition to important healthspan extension benefits.
Clinical Monitoring of Testosterone Optimization
Testosterone optimization should extend beyond symptom relief or achieving a target number. Whether using exogenous replacement or endogenous stimulation, therapy must be paired with structured laboratory monitoring to confirm effectiveness and safety.
Total and free testosterone establish treatment response. When using restoration strategies such as enclomiphene, luteinizing hormone, LH, provides important mechanistic insight. Unlike testosterone replacement therapy, which suppresses LH, successful endogenous stimulation should maintain or increase LH, confirming HPG axis engagement.
Because testosterone stimulates red blood cell production, hemoglobin and hematocrit require regular monitoring. Moderate increases are expected, but hematocrit approaching 52–54 percent warrants reassessment and potential dose adjustment [7]. Estradiol should also be followed, as excessive elevations may contribute to gynecomastia or mood changes. Markers such as SHBG and albumin help interpret free testosterone accurately and may reflect broader metabolic health.
Although not included with this review, aromatase inhibitors can increase testosterone by reducing estradiol-mediated feedback, routine estrogen suppression is generally not recommended in healthspan-focused care [8]. Estradiol plays important roles in bone density, vascular function, and metabolic regulation in men [8]. Rather than reflexively suppressing aromatization, addressing excess visceral adiposity and metabolic dysfunction is typically the more physiologically aligned strategy.
Thoughtful monitoring ensures that testosterone optimization supports physiologic resilience rather than simply elevating laboratory values.
Lifestyle as a Force Multiplier in Hormone Optimization
Laboratory monitoring ensures safety and effectiveness, but labs and prescriptions alone do not always equate to optimal results. Even the most precisely titrated pharmacologic strategy cannot compensate for persistent metabolic dysfunction. Functional testosterone deficiency is frequently rooted in excess visceral adiposity, insulin resistance, chronic stress, and poor sleep; factors that increase aromatization of testosterone to estradiol and blunt central hypothalamic signaling [7,8,10].
For this reason, testosterone restoration should be viewed as part of a broader physiologic strategy rather than a standalone solution. Visceral fat reduction decreases aromatase activity and estrogen burden [7,21]. Improved insulin sensitivity enhances hypothalamic responsiveness. Resistance training increases androgen receptor density and preserves lean mass. Sleep stabilization supports healthy pulsatile luteinizing hormone release [22]. These interventions do not merely complement therapy, they amplify endogenous signaling and may reduce required pharmacologic dosing over time.
In practice, men initiating enclomiphene often demonstrate rising morning testosterone within weeks, with fuller biochemical and symptomatic improvements emerging over eight to twelve weeks, particularly when body composition and metabolic markers are improving concurrently. Monitoring should therefore extend beyond testosterone alone. Trends in luteinizing hormone provide mechanistic confirmation, estradiol guides dose refinement, hematocrit ensures safety, and metabolic markers such as fasting insulin, lipid profile, and waist circumference reflect whether endocrine normalization is translating into biomarkers synonymous with healthspan extension.
Testosterone optimization is a dynamic system and partnership rather than a static intervention. Medication can enhance physiology, but lifestyle habits provide long-term stabilization. When these elements align, enclomiphene becomes not merely a testosterone enhancer, but a tool for restoring regulatory balance in service of long-term healthspan.
Testosterone Optimization For Healthspan
Testosterone optimization should not be mistaken for cosmetic enhancement or performance excess. At its core, it is the restoration of endocrine integrity. Testosterone within a mid-physiologic range supports preservation of lean muscle mass, metabolic flexibility, reduced visceral fat accumulation, bone density maintenance, erythropoietic balance, and cognitive resilience. These are not aesthetic endpoints; they are pillars of durable healthspan.
When low testosterone is corrected thoughtfully and monitored appropriately, particularly through strategies that preserve endogenous signaling such as enclomiphene, the objective shifts from hormone replacement to hormonal alignment. By stimulating the HPG axis rather than bypassing it, enclomiphene maintains intrinsic production, preserves fertility, and avoids the structural suppression associated with exogenous testosterone therapy. For men with secondary or functional hypogonadism, this represents a physiologically coherent approach, one that enhances vitality while respecting the architecture of the endocrine system.
Conclusion
Ultimately, testosterone optimization is most powerful when embedded within a broader foundation of metabolic health, resistance training, sleep optimization, and stress regulation. Pharmacologic stimulation alone does not create resilience; it amplifies the terrain upon which it acts. In this context, enclomiphene is best understood not as a shortcut, but as a strategic tool, one that, when paired with disciplined lifestyle alignment and informed clinical oversight, can support sustained performance, vitality, and long-term metabolic stability.
Healthspan is not extended by supraphysiological testosterone levels. It is supported by restoring systems to their intended balance.
- Santi, D., Spaggiari, G., Furini, C. et al. Temporal trends in serum testosterone and luteinizing hormone levels indicate an ongoing resetting of hypothalamic-pituitary-gonadal function in healthy men: a systematic review. J Endocrinol Invest 48, 2721–2734 (2025). https://doi.org/10.1007/s40618-025-02671-9
- Fraile-Martínez, Ó., Ortega, M. A., & García-Montero, C. (2026). Understanding the Secular Decline in Testosterone: Mechanisms, Consequences, and Clinical Perspectives. International Journal of Molecular Sciences, 27(2), 692. https://doi.org/10.3390/ijms27020692
- Rao, P. K., Boulet, S. L., Mehta, A., Hotaling, J., Eisenberg, M. L., Honig, S. C., Warner, L., Kissin, D. M., Nangia, A. K., & Ross, L. S. (2017). Trends in Testosterone Replacement Therapy Use from 2003 to 2013 among Reproductive-Age Men in the United States. The Journal of urology, 197(4), 1121–1126. https://doi.org/10.1016/j.juro.2016.10.063
- Comhaire, F., & Mahmoud, A. (2016). The andrologist's contribution to a better life for ageing men: part 1. Andrologia, 48(1), 87–98. https://doi.org/10.1111/and.12485
- Comhaire, F., & Mahmoud, A. (2016). The andrologist's contribution to a better life for ageing men: part 2. Andrologia, 48(1), 99–110. https://doi.org/10.1111/and.12489
- Comhaire F. (2016). Hormone replacement therapy and longevity. Andrologia, 48(1), 65–68. https://doi.org/10.1111/and.12419
- Zitzmann, M., Soave, A., & Bier, S. (2026). Functional testosterone deficiency in aging men: Clinical impact, diagnostic pathways, and treatment strategies. Maturitas, 207, 108870. Advance online publication. https://doi.org/10.1016/j.maturitas.2026.108870
- Walia, A., Coady, P., Sofia‐Hernandez, B., Gudlavalleti, H., El‐Achkar, A., Dunnam, S. E., ... & Khera, M. (2025). Testosterone Replacement, Where Are We in 2025?. Trends in Urology & Men's Health, 16(6), e70016. https://onlinelibrary.wiley.com/doi/10.1002/tre.70016
- Hackett G. I. (2025). Long Term Cardiovascular Safety of Testosterone Therapy: A Review of the TRAVERSE Study. The world journal of men's health, 43(2), 282–290. https://doi.org/10.5534/wjmh.240081
- Rojas-Zambrano, J. G., Rojas-Zambrano, A., & Rojas-Zambrano, A. F. (2025). Impact of Testosterone on Male Health: A Systematic Review. Cureus, 17(4), e82917. https://doi.org/10.7759/cureus.82917
- Nieschlag, E., & Nieschlag, S. (2019). ENDOCRINE HISTORY: The history of discovery, synthesis and development of testosterone for clinical use. European journal of endocrinology, 180(6), R201–R212. https://doi.org/10.1530/EJE-19-0071
- Grossmann, M., Robledo, K. P., Daniel, M., Handelsman, D. J., Inder, W. J., Stuckey, B. G. A., Yeap, B. B., Ng Tang Fui, M., Bracken, K., Allan, C. A., Jesudason, D., Zajac, J. D., & Wittert, G. A. (2024). Testosterone Treatment, Weight Loss, and Health-related Quality of Life and Psychosocial Function in Men: A 2-year Randomized Controlled Trial. The Journal of clinical endocrinology and metabolism, 109(8), 2019–2028. https://doi.org/10.1210/clinem/dgae085
- Hohl, A., Chavez, M. P., Pasqualotto, E., Ferreira, R. O. M., Sande-Lee, S. V., & Ronsoni, M. F. (2025). Clomiphene or enclomiphene citrate for the treatment of male hypogonadism: a systematic review and meta-analysis of randomized controlled trials. Archives of endocrinology and metabolism, 69(5), e250093. https://doi.org/10.20945/2359-4292-2025-0093
- Madhusoodanan, V., Patel, P., Lima, T. F. N., Gondokusumo, J., Lo, E., Thirumavalavan, N., Lipshultz, L. I., & Ramasamy, R. (2019). Human Chorionic Gonadotropin monotherapy for the treatment of hypogonadal symptoms in men with total testosterone > 300 ng/dL. International braz j urol : official journal of the Brazilian Society of Urology, 45(5), 1008–1012. https://doi.org/10.1590/S1677-5538.IBJU.2019.0132
- Rae J. M. (2011). Personalized tamoxifen: what is the best way forward?. Journal of clinical oncology : official journal of the American Society of Clinical Oncology, 29(24), 3206–3208. https://doi.org/10.1200/JCO.2011.36.3895
- Wibowo, E., Pollock, P. A., Hollis, N., & Wassersug, R. J. (2016). Tamoxifen in men: a review of adverse events. Andrology, 4(5), 776–788. https://doi.org/10.1111/andr.12197
- Saffati, G., Kassab, J., Orozco Rendon, D., Hinojosa-Gonzalez, D. E., Kronstedt, S., Lipshultz, L. I., & Khera, M. (2024). Safety and efficacy of enclomiphene and clomiphene for hypogonadal men. Translational andrology and urology, 13(9), 1984–1990. https://doi.org/10.21037/tau-24-238
- Helo, S., Mahon, J., Ellen, J., Wiehle, R., Fontenot, G., Hsu, K., ... & McCullough, A. (2017). Serum levels of enclomiphene and zuclomiphene in men with hypogonadism on long‐term clomiphene citrate treatment. BJU international, 119(1), 171-176. https://doi.org/10.1111/bju.13625
- Kim, E. D., McCullough, A., & Kaminetsky, J. (2016). Oral enclomiphene citrate raises testosterone and preserves sperm counts in obese hypogonadal men, unlike topical testosterone: restoration instead of replacement. BJU international, 117(4), 677-685. https://doi.org/10.1111/bju.13337
- Vermeulen, A., Goemaere, S., & Kaufman, J. M. (1999). Testosterone, body composition and aging. Journal of endocrinological investigation, 22(5 Suppl), 110–116. https://pubmed.ncbi.nlm.nih.gov/10442580/
- Zitzmann, M. Testosterone deficiency, insulin resistance and the metabolic syndrome. Nat Rev Endocrinol 5, 673–681 (2009). https://doi.org/10.1038/nrendo.2009.212
- Pastuszak, A. W., Moon, Y. M., Scovell, J., Badal, J., Lamb, D. J., Link, R. E., & Lipshultz, L. I. (2017). Poor Sleep Quality Predicts Hypogonadal Symptoms and Sexual Dysfunction in Male Nonstandard Shift Workers. Urology, 102, 121–125. https://doi.org/10.1016/j.urology.2016.11.033